

KEY WORDS
Clinical care, corona, Covid-19
INTRODUCTION
On February 3rd, 2020, the first case of SARS-CoV-2 (Severe acute respiratory syndrome – Coronavirus-2) infection was diagnosed in Belgium in an asymptomatic patient who was quarantined after evacuation from Wuhan, China. On February 29th, the second Belgian patient was admitted to the Antwerp University Hospital after travel from France.1 Since then, there was a rapid rise in cases, with 13964 confirmed cases, 6132 admissions, and 828 deaths as of April 1st. 2 Limburg Province has become the epicentre of the Belgian outbreak with the highest incidence across the country. Jessa Hospital is a 981-bed non-academic tertiary care centre, located in the centre of the Limburg Province in Belgium. In our hospital, the first diagnosis of the coronavirus disease-2019 (Covid-19) was made on March 3rd in an ambulatory patient and the first patient was admitted on March 11th. As of April 1st, a total of 235 patients had been admitted to our hospital of whom 72 were discharged, 38 died, and 125 are still in the hospital (figure 1). In this article, we would like to share our experience in the handling of some of the practical and organisational issues in the care for Covid-19 patients.
ORGANISATIONAL MEASURES
We started preparing for the upcoming outbreak six weeks before the first patient was admitted, when an outbreak management team was installed. Since then, we encountered many challenges in the organisation and care for Covid-19 patients. In a timeframe of only a few weeks, a large hospital had to be transformed into a ‘Covid’ hospital that would be able to treat a large number of Covid-19 pneumonia patients, while at the same time, be able to ensure safe urgent care for non-Covid-19 patients. Close follow-up of data and information from China, Singapore, Italy, and other affected countries was paramount in this organisational transformation. Supported by governmental measures, we cancelled all consultations, ambulatory surgery, and elective admissions.
We implemented general principles for hospital disaster preparedness from the beginning. As stated in the official Belgian emergency plan, the Chief Medical Officer (CMO) of the hospital was granted expanded authority to implement the measures needed to cope with the epidemic. In the deployment of admission capacity, a ratio of 1:4 (ICU : non-ICU patients) was used to estimate the intensive care capacity needed. We increased the number of intensive care unit (ICU) beds from 48 to 60 (and in extreme conditions expandable to 70) by transforming a large part of our operating rooms into an ICU. Since the Jessa Hospital is a reference centre for cardiology and oncology, we were able to convince the authorities that 40% of our full intensive care capacity needed to be reserved for non-Covid-19 patients such as, for example, urgent cardiac surgery and surgical oncology.
Our experience so far shows that pro-active communication of planned organisational measures is of crucial importance. In a daily newsletter, full transparency was created to all physicians about the hospital metrics of COVID-19 and the national and international evolution of the epidemic, as well as all organisational adaptations needed to cope with the expected surge of Covid-19 patients. In addition, the press officer of the hospital informed both the local and national media from the beginning of the numbers of admissions, deaths, and discharged COVID-19 patients.
In the implementation of the transformations of the emergency department (ED), ICU, and multiple standard wards towards COVID-wards, our general principle was to stay at least one week ahead of the epidemic impact. In reality, this provided a timeframe of several days in which both physicians and nurses could adapt to their new ward and receive education on, for example, the use of protective measures and other protocols before the first patients were admitted.
As much as possible, existing teams of physicians and of nurses were kept together on the new wards. At the start of deployment, pulmonologists, geriatric physicians, and infectious disease specialists were responsible for to the first wards. Quite rapidly, all specialists in internal medicine of the hospital were appointed to a specific Covid-19 ward. Since all elective surgery was cancelled, we were able to integrate the majority of anaesthesiologists within the team of intensive care specialists to supervise all Covid-19 ICUs. We also reserved part of a paediatric ward for Covid-19 and created a small unit for Covid-19 postnatal care.
In the final stage of deployment, these teams of internal medicine specialists – taking into account that we expected up to 20% of physicians to be affected by Covid-19 and would be on sick leave for 1-2 weeks - were supported by a backup of specialists from surgical disciplines.
Empirically, the ratio of geriatric versus non-geriatric wards was about 1:1. Nightly permanence on-site had to be doubled for nurses on the wards, and tripled for intensive care specialists for the Covid-19 ICUs and for supervision of the multiple regular Covid-19 wards.
Notably, we were able to control the number of inappropriate referrals to the ED by establishing an integrated care Covid-19 protocol with local general practitioner (GP) circles. The local GPs created and manned a specific Covid-19 referral centre for GPs, located next to the hospital. Based on elaborate ethical triage measures, admission policy from local residential care homes for Covid-19 intensive care was also highly restricted.
ORGANISATION OF THE CLINICAL PATIENT FLOW
From the start of the epidemic, the ED was separated into a corona unit and a corona-free unit. Patients with possible Covid-19 were seen by emergency physicians in a separate location within the ED department where three linked rooms provided a waiting space, a patient assessment room with a negative-pressure system in place, and a dressing room for personnel. From there, the patient was admitted to an isolation negative-pressure room in the Pulmonology Department while awaiting test results. Soon it became clear that the influx of patients would outnumber the capacity of the ED and the pulmonology ward. Therefore, on March 13th two transit wards were opened with 20 beds each. All patients who were triaged in the ED with possible Covid-19 based on clinical assessment, arterial blood gas test, and indication for hospital admission were tested for SARS-CoV-2 by nasopharyngeal swab and subsequently admitted to one of the transit departments for further clinical assessment while awaiting test results. Patients with a geriatric profile, defined as over 75 years of age and/ or with multiple comorbidities were admitted to a ward supervised by a geriatric specialist. All other patients with possible Covid-19 virus were admitted to a ward supervised by an infectious disease specialist. As soon as the test results became available, the patient was transferred to a non-Covid-19 ward in cases of a negative results or to a dedicated Covid-19 ward in cases of a positive result. In addition, in the geriatric corona ward a section of eight beds was reserved for palliative care.
Initially, patients with suspected Covid-19 who presented at the ED and who needed immediate intensive care or cardiac monitoring were directly transferred to a dedicated corona ICU unit (with 14 beds) without awaiting the SARS CoV-2 PCR results. However, to prevent nosocomial transmission of Covid-19 to patients with a Covid-19-negative test, one intensive care room (‘crash box’) was created at the non-geriatric transit ward where intubation, ventilation, vasopressive support, and cardiac monitoring could be performed while awaiting test results. In addition, for patients who needed emergency surgery for any reason and were Covid-19 positive, a dedicated surgical ward was opened for optimal post-surgical care. This ward also functioned as a post-intensive care rehabilitation unit. With the increase in Covid-19-patients, subsequential Covid wards were opened. On April 1st, a total of 10 Covid wards and 5 ICU wards were operational (figure 2). This resulted in a total capacity of 200 Covid beds including 41 ICU beds. 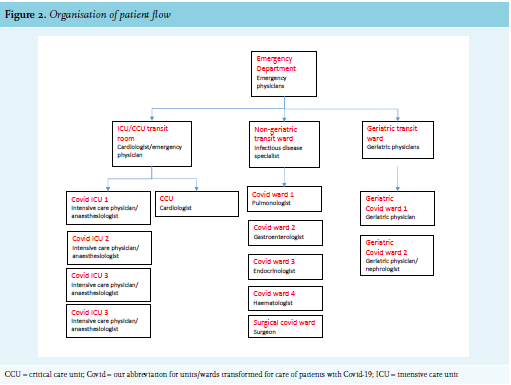
We noted a remarkable similar preadmission disease course in most patients, which reflects the first reports from China.3,4 The disease starts with mild respiratory symptoms, fever, and malaise. During the course of 5-8 days there is a gradual progressive dyspnoea. Although during the course of the epidemic, it became clear that a significant portion of patients present with atypical symptoms such as anosmia, abdominal pain, vomiting, and diarrhoea.5,6 Triage criteria in the ED were adjusted accordingly. Findings in laboratory evaluations that we see also reflect the first clinical reports.6,7 The combination of elevated lactate dehydrogenase (LDH), elevated ferritin, a decreased partial pressure of oxygen (pO2) and partial pressure of carbon dioxide (pCO2) in arterial blood gas, and lymphocytopenia is almost universal. The C-reactive protein (CRP) levels may vary considerably. Nasopharyngeal swabs were analysed with an in-house developed reverse-transcriptase PCR for the E-gene on the ARIES analyser (Luminex Corporation). Test results were available within three to four hours. Additionally, a chest X-ray was performed for all suspected cases.
A dilemma arises in patients with a high clinical suspicion and a negative PCR test result. The exact sensitivity of the test depends on the location where the swab was taken and the duration of symptoms, and is obviously influenced by the sampling technique of the person collecting the swabs.8 Training of personnel in correct sampling technique was therefore provided with online instructional videos.
In patients with a clinical suspicion based on history, laboratory and/or chest X-ray results, and a negative initial PCR test, the following strategy was adopted: a second nasopharyngeal swab was taken after 24-48 hours. If it is possible to collect a deeper sample such as sputum, the PCR is repeated on this sample. If a patient is intubated, a lower respiratory tract aspirate is collected, as this has been shown to be more sensitive than a nasopharyngeal swab.8,9 In the second sample, additional screening for other viral pathogens and atypical bacterial pathogens by PCR is performed. While awaiting the second test, a urinary antigen test for Streptococcuspneumoniae and Legionella species is done, and additional serological testing for HIV is performed. When the additional investigations are all negative, a chest computed tomography (CT) scan is performed to evaluate for typical signs of viral pneumonitis (peripheral ground-glass opacities and/or mixed consolidations).10 If the CT scan is suggestive of COVID-19 and no other diagnosis is established, the patient is considered as having COVID-19. During all these investigations, the patient remains in isolation in the transit wards.
To date, we have identified 10 patients where a second nasopharyngeal swab tested positive after a first negative test. In addition, we saw one patient with two negative nasopharyngeal swabs, who tested positive on a lower respiratory tract aspirate taken after intubation. Studies are needed in order to develop the best strategy in these situations.
THERAPY
As there is currently no specific evidence-based antiviral therapy for Covid-19, patients were treated with optimal supportive care, including oxygen therapy.
Covid-19 stewardship team
To support the physicians on the Covid-19 wards and to guarantee a uniform approach throughout the hospital, we installed a Covid-19 stewardship team. This team includes an infectious disease specialist, a pulmonologist, a clinical microbiologist, and a clinical pharmacist. The team visits the different Covid-19 wards and ICU twice a week to discuss any issues on therapy and other patient care with the treating physician. In addition, a hospital clinical guideline containing all Covid-19 related procedures was available through the hospital intranet and distributed actively to all physicians supervising Covid units. This document continues to be updated twice a week.
Hydroxychloroquine
Patients were offered hydroxychloroquine if they met the criteria defined by the Belgian national guideline for treatment of Covid-19.11 Preferably, an echocardiogram (ECG) was performed before the start of hydroxychloroquine treatment to rule out QTc prolongation. However, we decided that in patients without risk factors for QTc time prolongation (history of cardiovascular disease, bradycardia < 50/min, hypokalaemia, hypomagnesaemia, renal insufficiency, hepatic insufficiency, other QTc prolonging drugs) hydroxychloroquine could be started and the ECG was to be performed the next day. In patients with a QTc > 500 ms, hydroxychloroquine is not started or is stopped. In patients with a QTc between 450 and 500 ms, a daily ECG was conducted. If QTc time is < 450 ms, there is no follow-up ECG.
Empiric antibiotic therapy
Because little was known about possible bacterial superinfection, we initially decided to start empiric antibiotic therapy with amoxicillin-clavulanic acid for five days in all patients with pulmonary infiltrates on chest X-ray. However, we changed this policy because, based on our clinical judgement, antibiotics were unnecessary in many patients. In addition, we noticed that in patients admitted to the ICU, there was a tendency of early empirical switch to broader spectrum antibiotics such as piperacillintazobactam in cases of clinical deterioration. After a consultation round with our infectious disease specialists, pulmonologists, clinical microbiologists, and intensive care physicians we reached a consensus to limit empirical antibiotics to patients with signs of bacterial pneumonia. This was arbitrarily defined as patients with at least one of the following: leucocyte count > 15,000 *106 /l, cough productive with sputum, or a lobar infiltrate on radiology.
Prevention of thromboembolism
Covid-19 is a pro-thrombogenic condition. A recently published study showed that therapeutic doses of anticoagulation with low molecular weight heparin (LMWH) decreases mortality in patients who need ICU care and in patients with a D-dimer of > six times the upper limit of normal.12 We give all our patients with Covid-19 LMWH in prophylactic dosage unless there is a clear contraindication or if the patient already uses therapeutic anticoagulants. Covid-19 is a pro-thrombogenic condition. A recently published study showed that therapeutic doses of anticoagulation with low molecular weight heparin (LMWH) decreases mortality in patients who need ICU care and in patients with a D-dimer of > six times the upper limit of normal.12 We give all our patients with Covid-19 LMWH in prophylactic dosage unless there is a clear contraindication or if the patient already uses therapeutic anticoagulants.
Fluid resuscitation
Acute respiratory distress syndrome (ARDS) is a frequent complication of Covid-19 with high mortality.3,4,6 Previous studies have shown that in general, restrictive fluid policies prevent ARDS in intensive care patients.13 On the other hand, many ICU patients develop acute tubular necrosis.14 Therefore, it is a delicate balance to restrict fluid administration but also prevent hypovolaemia.
Bronchodilators
Bronchospasm is infrequent in our patients without history of bronchial asthma or COPD. In patients who do need bronchodilators, we try to give inhalation medication only through a dose inhalator with spacer to prevent excessive exposure of health care workers to contaminated aerosols.
Psychosocial support team
A Corona support team was formed to support patients and families in addition to the regular support provided by medical staff. The Corona support team consists of a psychiatrist and psychologist who visit the Covid-19 wards at fixed hours on a daily basis. In addition to psychosocial support to patients and families, the Corona support team is also available for psychological support of health care professionals.
DISCHARGE AND FOLLOW-UP
Initially, we noticed a reluctance to discharge patients who were hospitalised in a ward supervised by physicians who have less experience with patients with pneumonia because of fear of late clinical deterioration. In our experience, however, sudden clinical deterioration is unusual in patients who show a consistent clinical improvement. We have adopted the following rule of thumb to discharge a patient: All patients who do not need oxygen for 24 hours AND have a sO2 ≥ 94% AND no apparent respiratory distress AND temperature < 38°C in last 48 hours are discharged. In addition, patients with a clear clinical improvement who need ≤ 2 l O2 /min are also discharged with home oxygen supply. All patients are instructed to contact the hospital in case of clinical deterioration. As of April 1st only 3 of 72 discharged patients have been readmitted because of late clinical deterioration.
CONCLUSION
We have faced many challenges with the onset of the Covid-19 outbreak in our region. The situation is constantly evolving with continuing great impact on our health care system. The extraordinary transformation of the hospital organisation and multiple care processes was undertaken using principles of hospital disaster preparedness with, as much as possible, pro-active deployment of hospital capacity, in an attempt to stay ahead of a possible collapse of the local health care system. In our experience, it has been useful to install transit wards where patients wait for their SARS-CoV-2 PCR test results. This prevents an overflow of the ED and unnecessary contact between Covid-19-negative and Covid-19-positive patients. In addition, because of the scale of the outbreak, physicians with different specialities are currently involved in the care of Covid-19 patients. This complicates a uniform policy for admitted patients. We have adopted a hospital-wide guide on the care of Covid-19 patients and have installed a multi-disciplinary Covid-19 stewardship team. A clear internal and external communication policy proved to be paramount to ensure an unforeseen organisational transformation of this large tertiary hospital.
In this paper, we have shared our experiences and we hope that this may be useful for other hospitals during the current crisis.
REFERENCES